
Will not because bromine is below fluorine on the periodic table. The elements on top of the column will replace the elements below them on the periodic table but not the other way around. Collectively, these elements are called the halogens and are in the next-to-last column on the periodic table (see Figure 4.1 “Halogens on the Periodic Table”). This is most easily demonstrated with fluorine, chlorine, bromine, and iodine. Not all proposed single-replacement reactions will occur between two given reactants. A typical characteristic of a single-replacement reaction is that there is one element as a reactant and another element as a product. Here the negatively charged ion changes from chloride to fluoride. Another example of a single-replacement reaction is The hydrogen atoms in HCl are replaced by Zn atoms, and in the process a new element-hydrogen-is formed. Is an example of a single-replacement reaction.

Here we will begin our study of certain types of chemical reactions that allow us to predict what the products of the reaction will be.Ī single-replacement reaction is a chemical reaction in which one element is substituted for another element in a compound, generating a new element and a new compound as products. Up to now, we have presented chemical reactions as a topic, but we have not discussed how the products of a chemical reaction can be predicted.
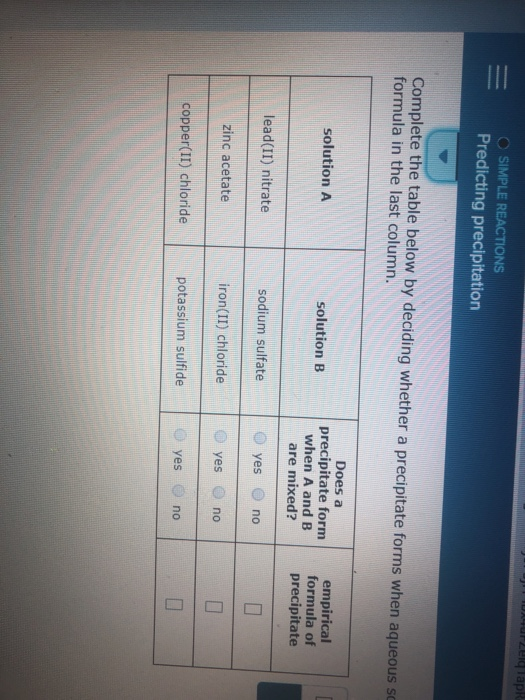
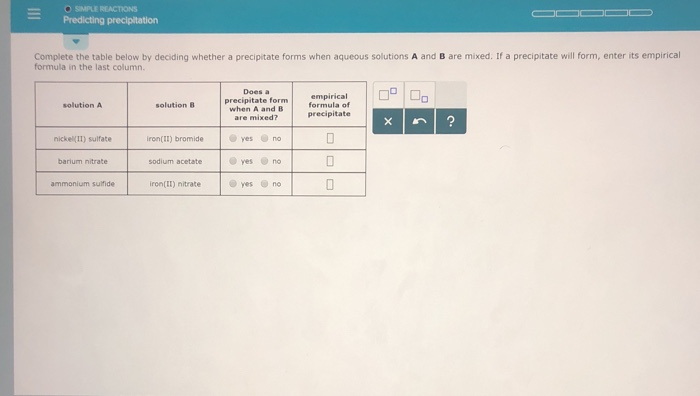
Recognize chemical reactions as single-replacement reactions and double-replacement reactions.Therefore, you can say that two precipitates will form when you mix the two solutions. In this case, the solubility product quotient will be equal to So a precipitate of strontium sulfate will form. #Q_(sp) > K_(sp) -># a precipitate will form until #Q_(sp) = K_(sp)#.The solubility product quotient, #Q_(sp)#, is calculated using the concentrations of the ions in the resulting solution.
# = (2 xx "0.2 M")/2 = "0.2 M"#įor strontium sulfate, the #K_(sp)# will look like this Since the volume of the solution doubles, the concentrations of the ions will be halved. You can calculate the concentration of the ions involved in the reaction after the two solutions are mixed. Keep in mind that strontium hydroxide is only slightly soluble in water, but its dissociation will be aided by the fact that the hydroxide anions will form a precipitate with the copper(II) cations. More specifically, this reaction will produce copper(II) hydroxide, #"Cu"("OH")_2#, and strontium sulfate, #"SrSO"_4#, which will precipitate out of solution. The interesting thing about this reaction is that both products are considered insoluble in aqueous solution.
